Development of a xenogeneic vascularized endocrine pancreas
16.02.2023
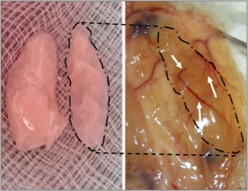
Intrahepatic islet transplantation is the standard cell therapy for β cell replacement. However, the shortage of organ donors and an unsatisfactory engraftment limit its application to selected patients with type 1 diabetes. There is an urgent need to identify alternative strategies based on an unlimited source of insulin producing cells and innovative scaffolds to foster cell interaction and integration to orchestrate physiological endocrine function. Previously the use of decellularized lung as a scaffold for β cell replacement was proposed with the final goal of engineering a vascularized endocrine organ. In collaboration with the group of Antonio Citro and Lorenzo Piemonti in Milano, this technology was prototyped by integration of neonatal porcine islets and healthy subject-derived blood outgrowth endothelial cells to engineer a xenogeneic vascularized endocrine pancreas. This technology not only is able to foster neonatal pig islet maturation in vitro, but also to perform in vivo immediately upon transplantation and for over 18 weeks, compared to normal performance within 8 weeks in various state of the art preclinical models. Given the recent progress in donor pig genetic engineering, this technology may enable the assembly of immune-protected functional endocrine organs.